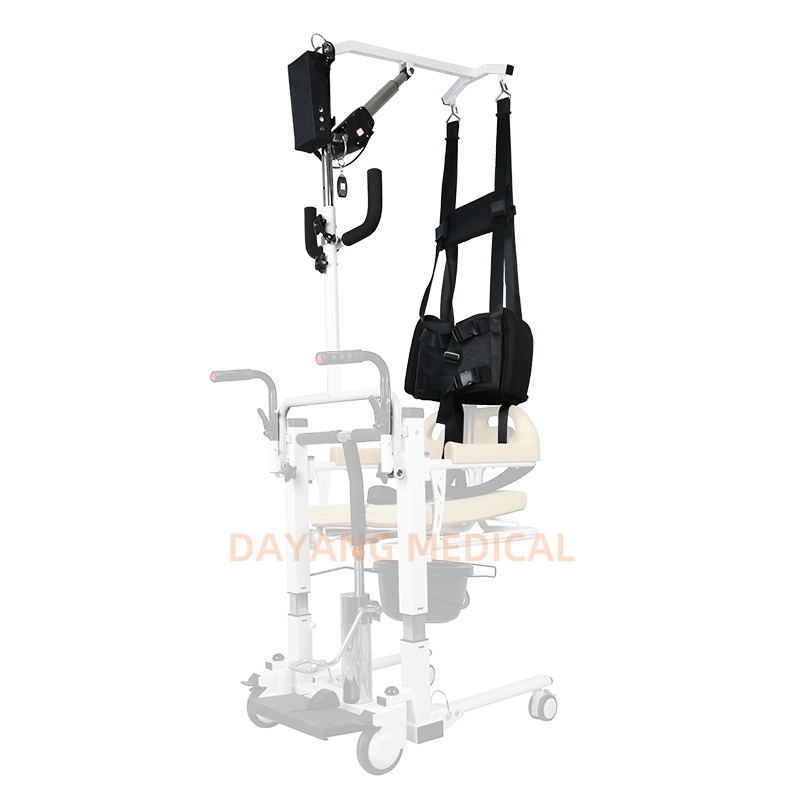
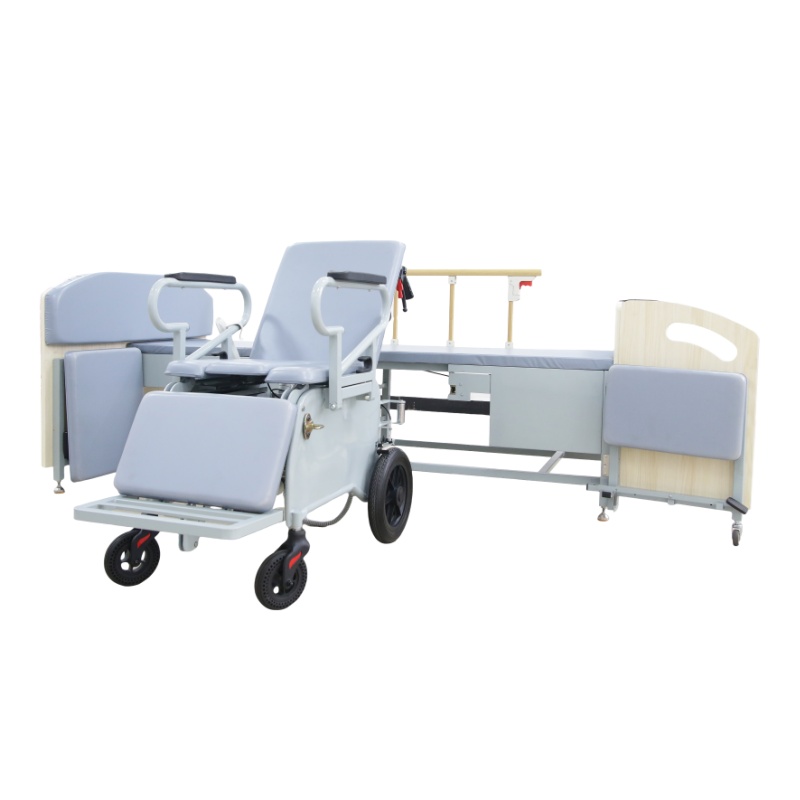
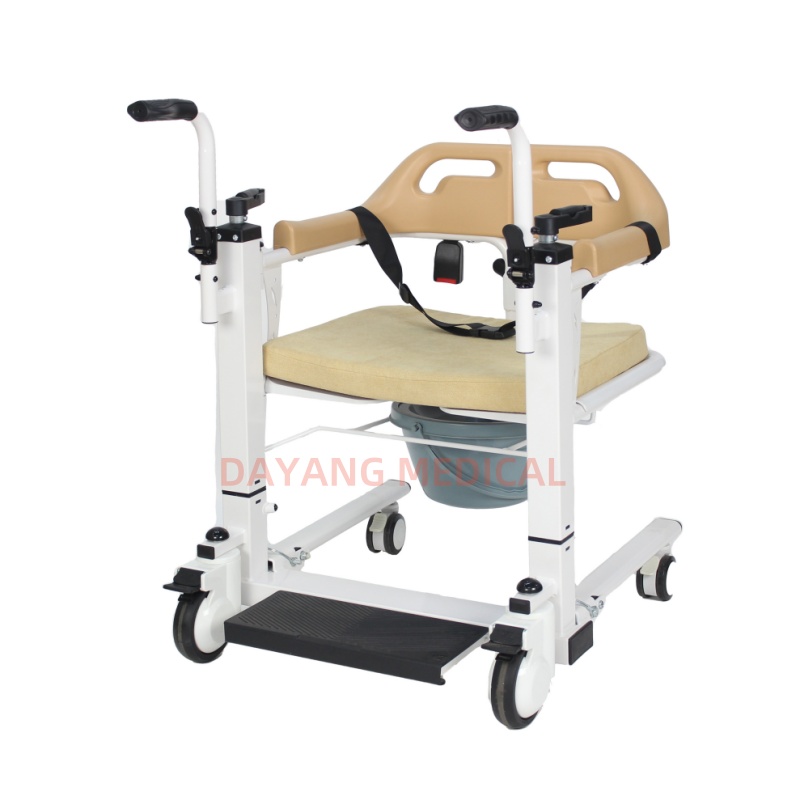
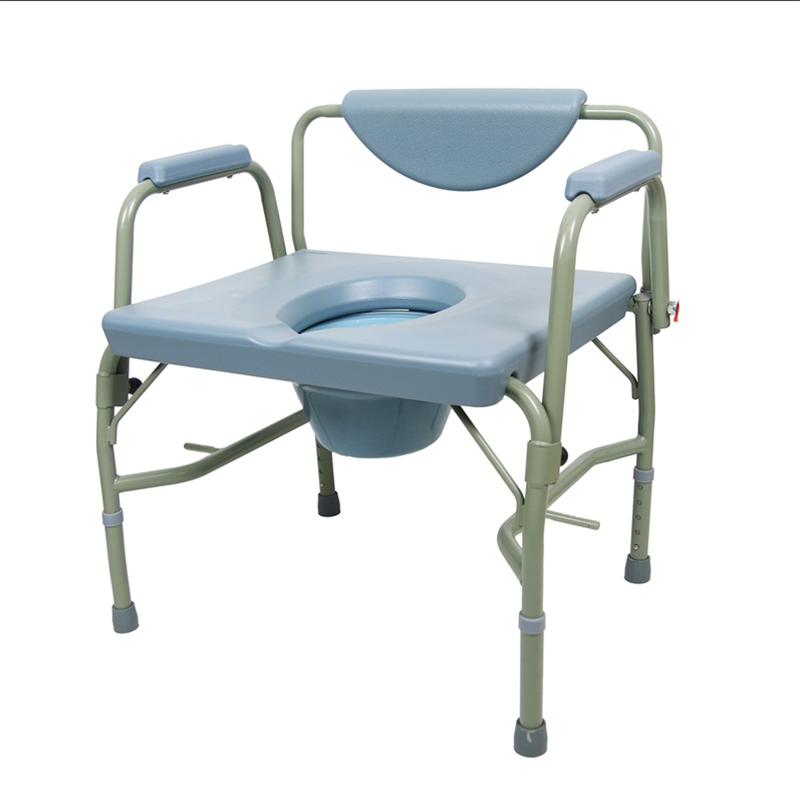
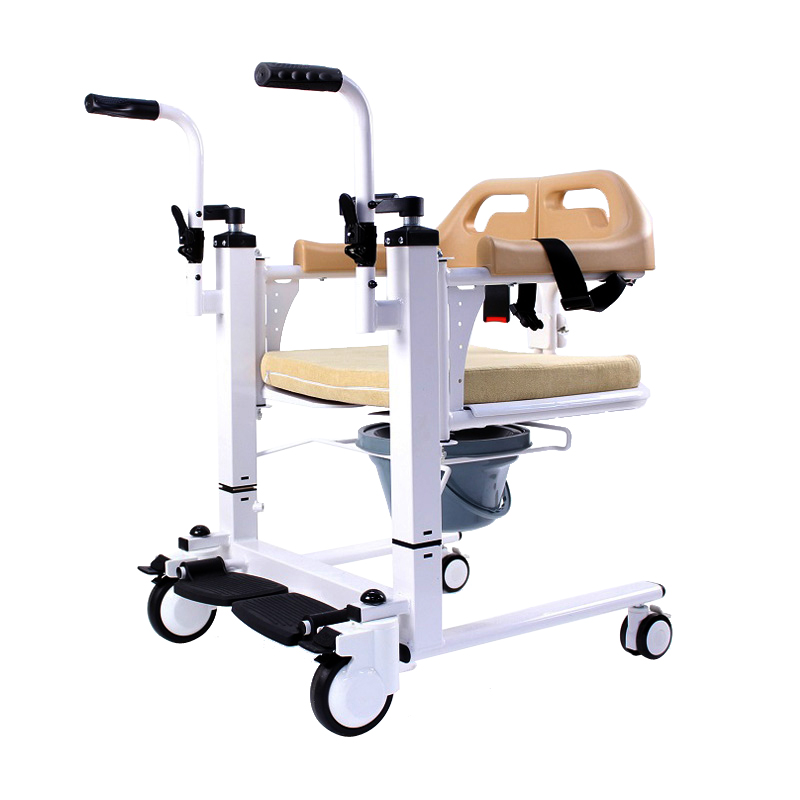
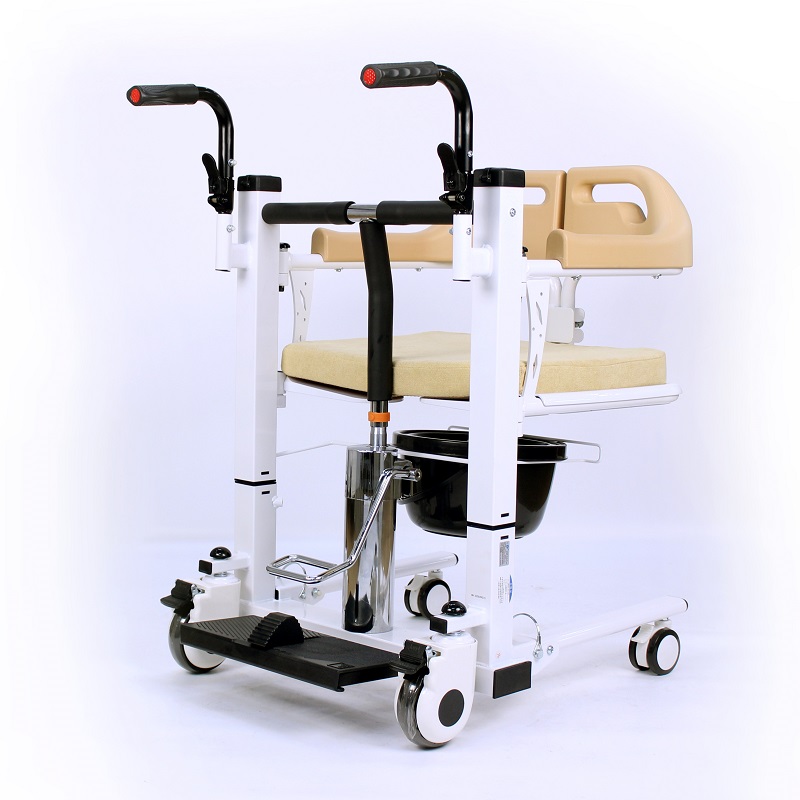
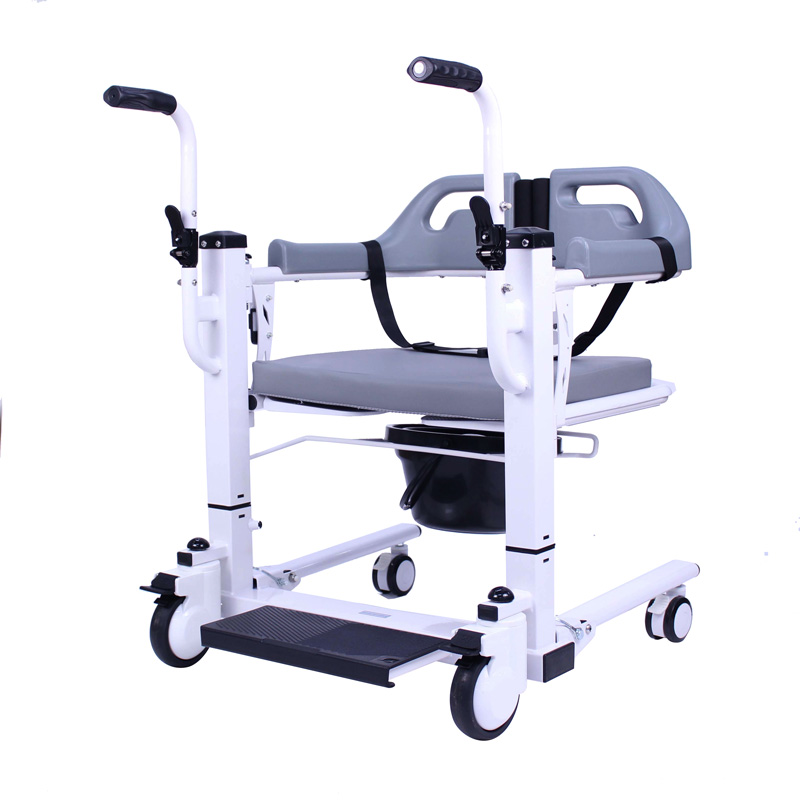
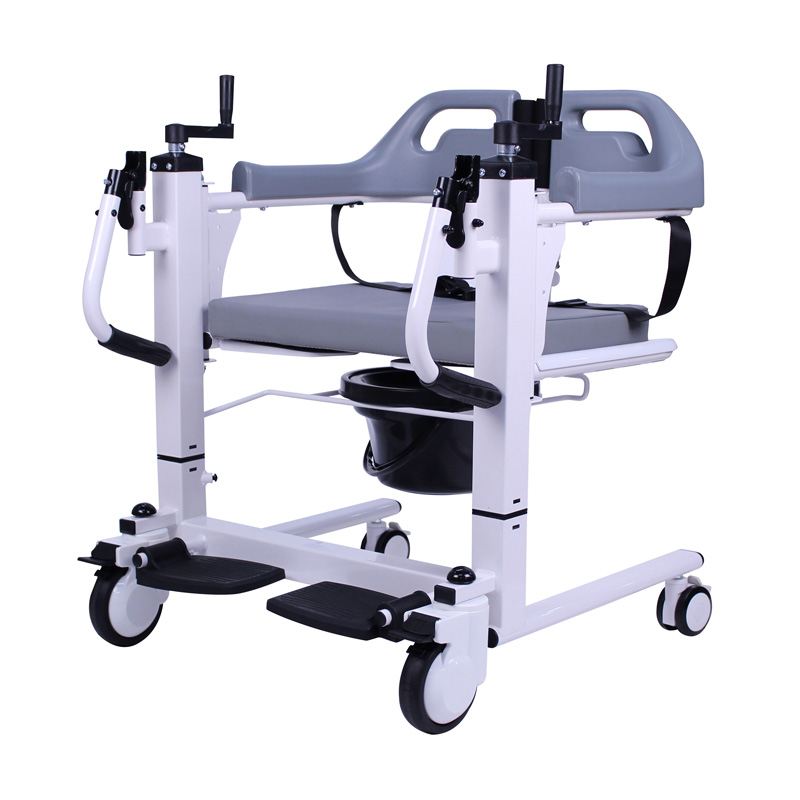
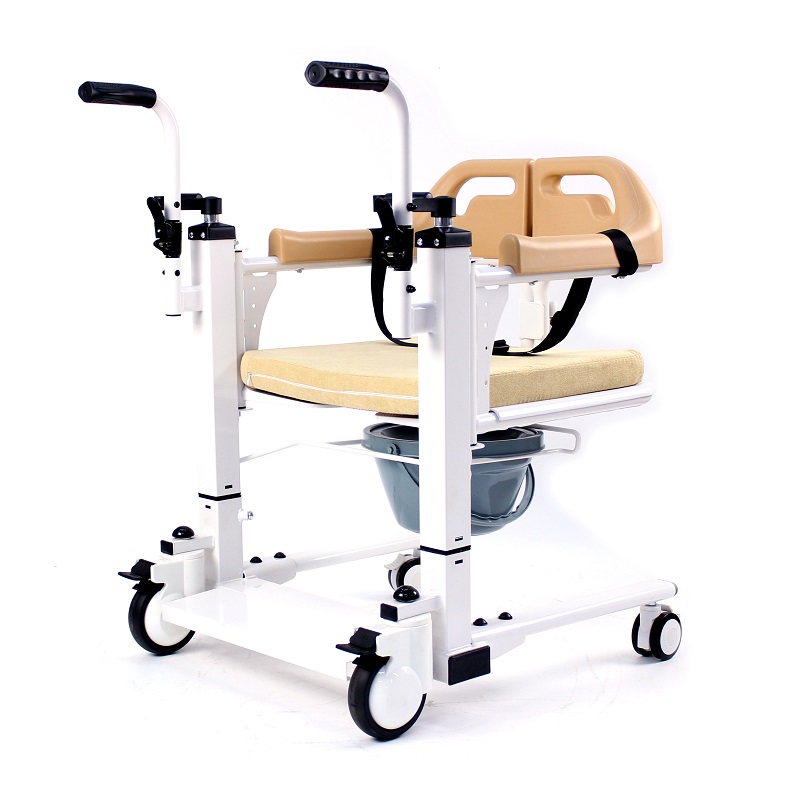
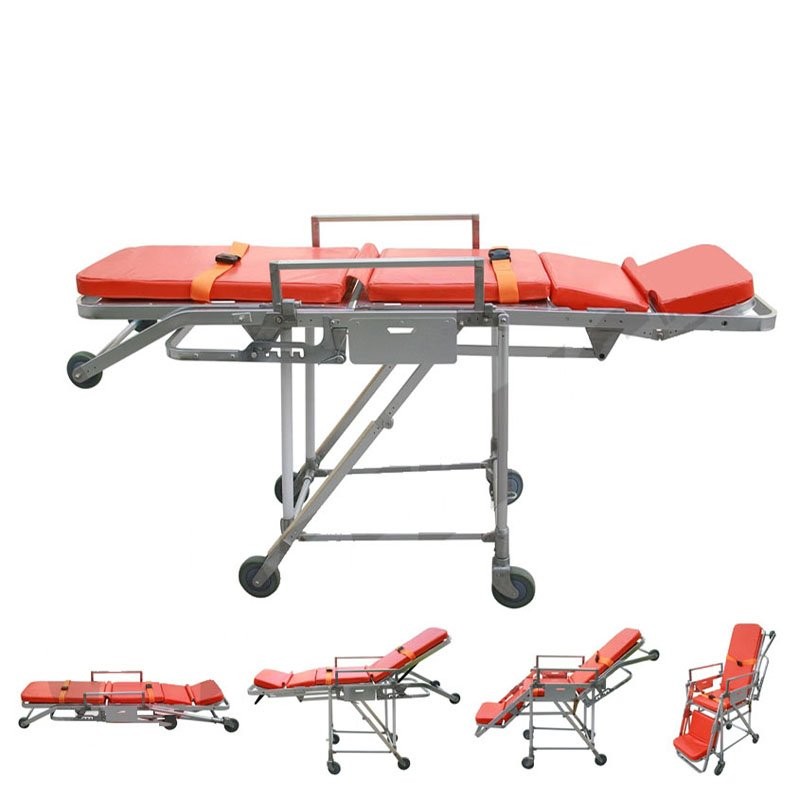
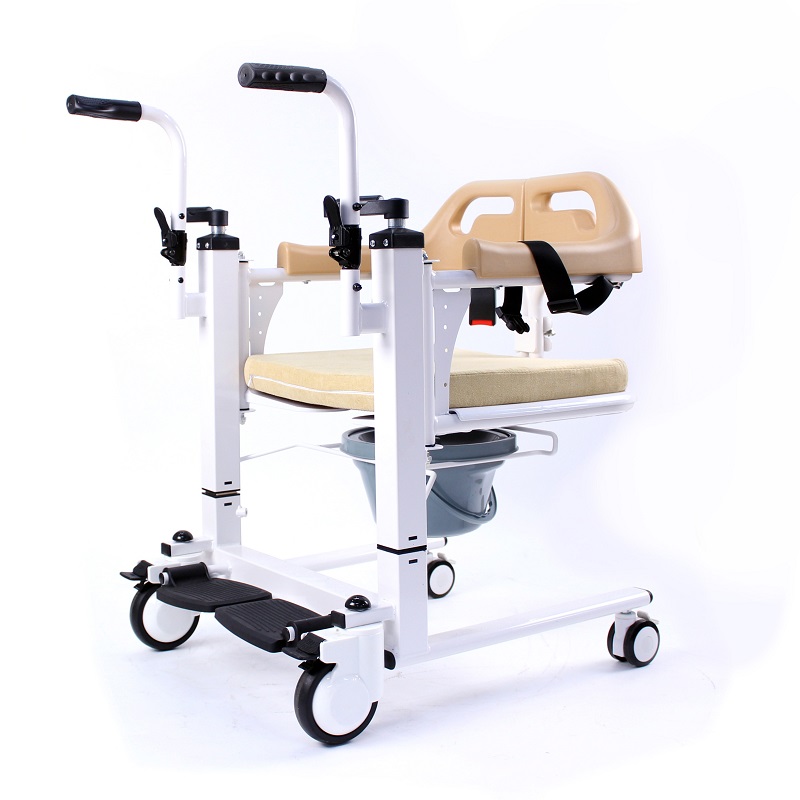
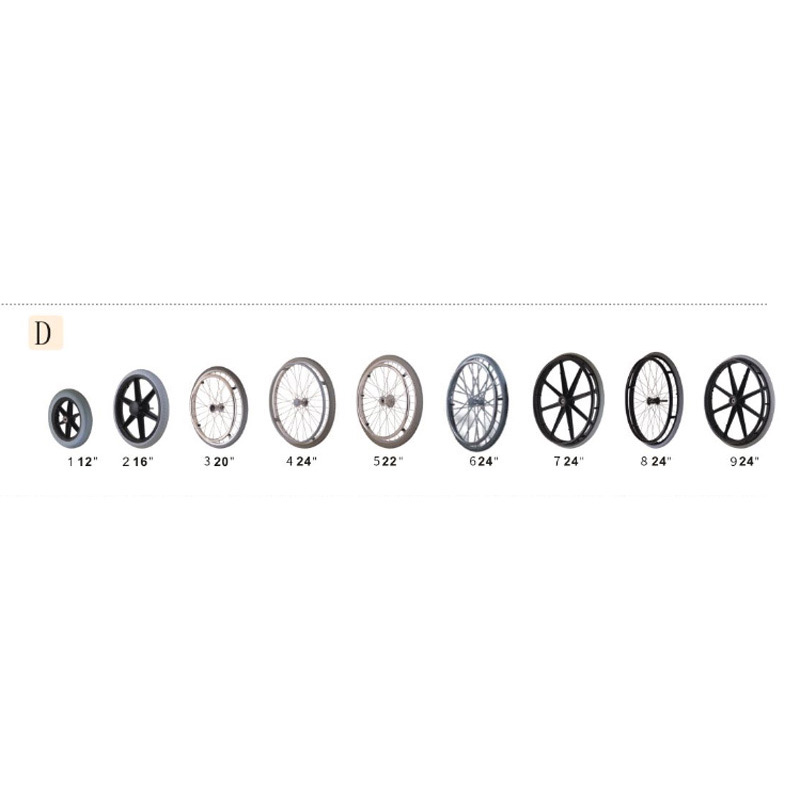
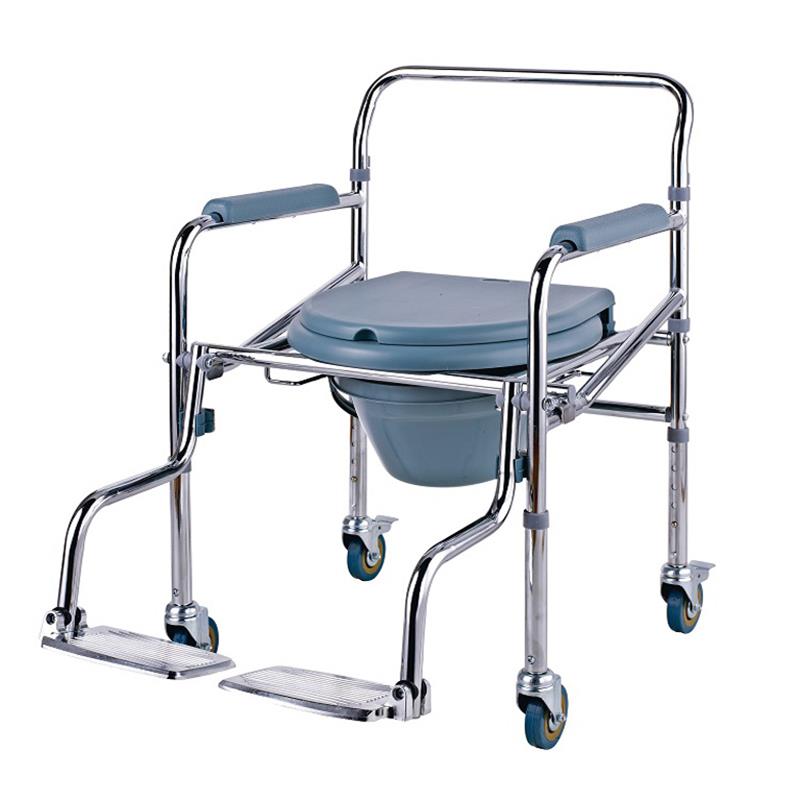
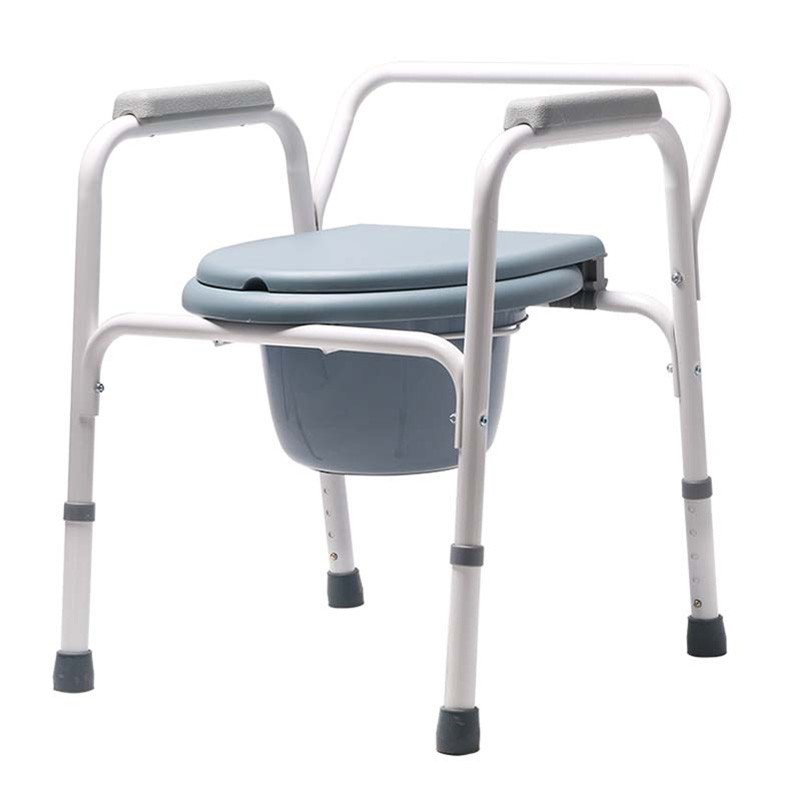
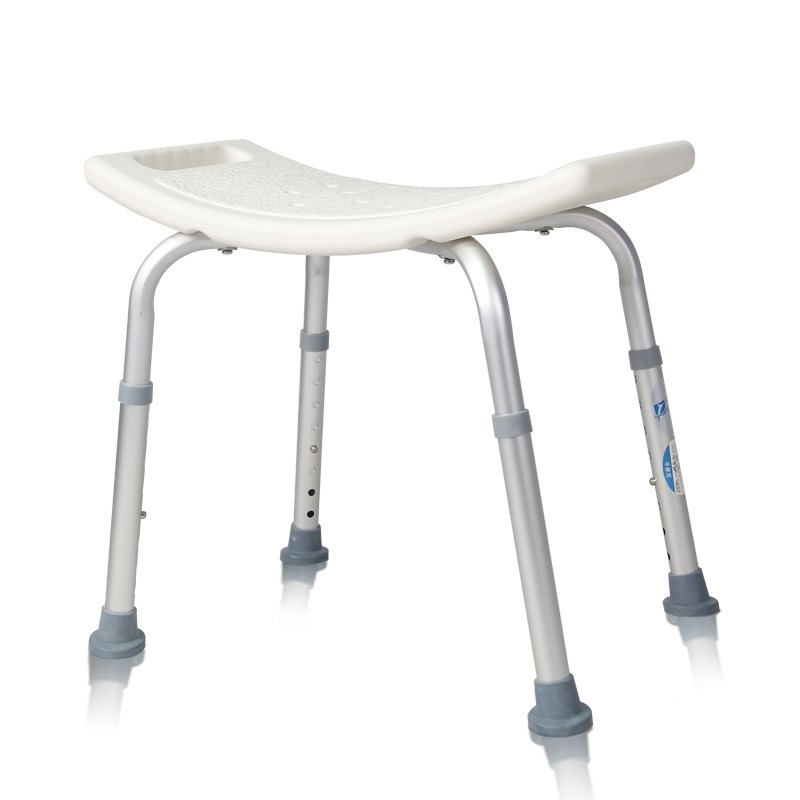
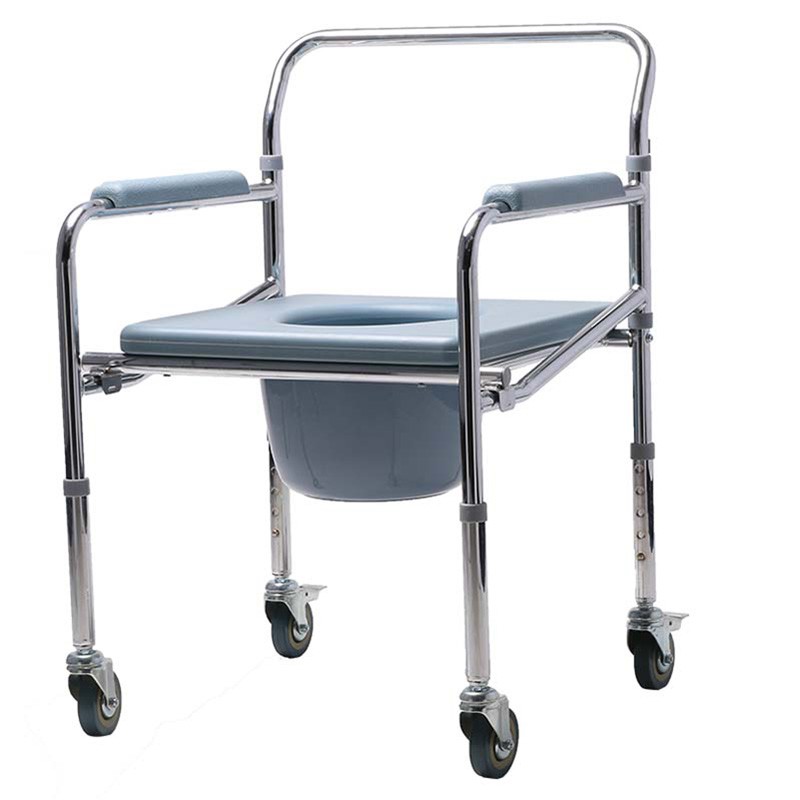
The Certificate For Exportation of Medical Products are certified to be in compliance with the standards and regulations of the People's Republic of China regarding medical devices, registered in China and authorised for sale in the Chinese market. There is no restriction on the export of the product.
This certificate is used to certify the legality of the sale of the above product in the People's Republic of China and serves as the basis for the export of the product.
Issued and approved by Guangdong Provincial Drug Administration.